|
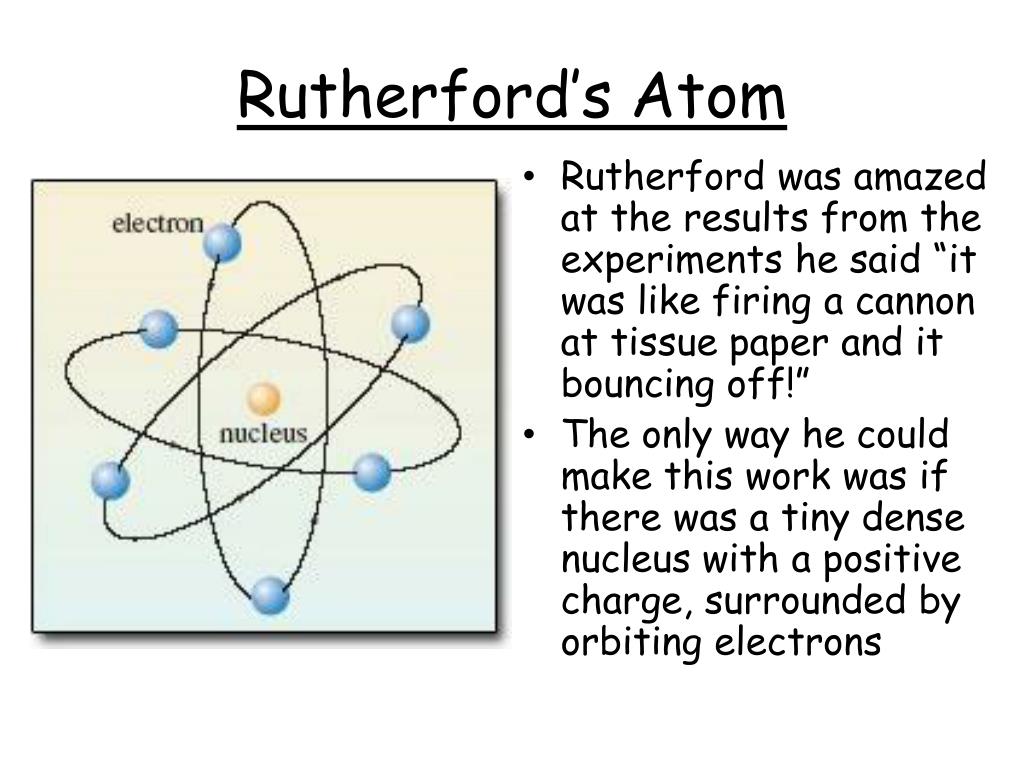
AND if they already knew that the electron was small and negative, then the atom must have a small positive nucleus with the electrons around them. If the positive alpha particles mostly passed through the foil, but some bounced back. How could that be if the plumb pudding model was correct? Rutherford's experiment prompted a change in the atomic model. Rutherford found that most of them went right through the foil. If you shoot these positive alpha particles at this positive pudding atom, they should mostly bounce off, right? Well, that is not what happened. He shot some alpha particles (which are really just the nucleus of a helium atom) at some really thin gold foil. According to classical theory, electrons moving on an orbit should emit electromagnetic radiation and the electrons would therefore lose energy and spiral into the nucleus. This module takes readers through experiments with cathode ray tubes that led to the discovery of the first subatomic particle: the electron. The Bohr atomic model After working with both Thomson and Rutherford, Niels Bohr, in 1913, realised that Rutherfords model was unstable. One of his best-known students was Ernest Rutherford, who succeeded Thomson as Cavendish Professor of Physics.Ernest Rutherford said one day "hey, I think I will shoot some stuff at atoms." I am sure his wife said "oh, Ernie" (she probably called him Ernie) "if it makes you happy to play with your little physics stuff, go ahead. The 19th and early 20th centuries saw great advances in our understanding of the atom. Seven of his research assistants, as well as his own son, went on to win the Nobel Prize in Physics. Thomson's greatest contribution to science to be his role as a teacher. Thomson published an important monograph in 1913 urging the use of the mass spectrograph in chemical analysis. His atomic theory helped explain atomic bonding and the structure of molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
